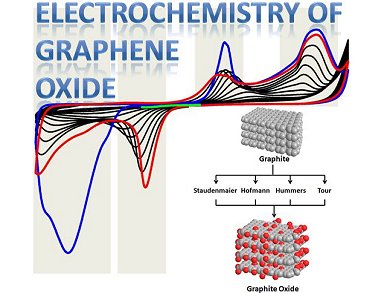
Graphene oxides are materials of significant interest for the use in electrochemical devices. These materials are typically prepared by oxidizing graphite in strong mineral acid mixtures with chlorate or permanganate oxidants. Martin Pumera and his group, Nanyang Technological University, Singapore, in collaboration with a group from Prague, Czech Republic, have discovered that the oxidant used has a profound effect on the electrochemistry of the resulting graphene oxide. Materials prepared with chlorate exhibit chemically irreversible reductions, whereas those prepared with permanganate have very unusual inherent chemically reversible electrochemistry of their oxygen-containing groups.
By combining precise electrochemical pretreatments with XPS analysis, they were able to establish that the use of permanganate oxidants generates distinctive oxygen functional groups. Specifically, they noted the increased presence of carbonyl and carboxyl groups on graphene from oxidation processes. A pH study indicates that quinone–hydroquinone electrochemistry are likely sources of the reversible character observed for these graphene oxides.
Hence, such graphene oxides with controllable redox states have potential as transducers in electrochemical and biosensing applications.
- Unusual Inherent Electrochemistry of Graphene Oxides Prepared Using Permanganate Oxidants,
A. Y. S. Eng, A. Ambrosi, C. K. Chua, F. Šaněk, Z. Sofer, M. Pumera,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201301889