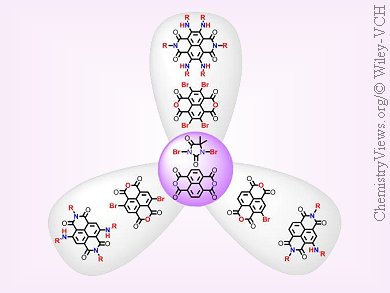
Functionalized naphthalene diimides (NDIs) are potential building blocks to bridge the gap between molecular and macroscopic worlds because of their potential applications that range from electronics and biomedicine to nanotechnology. The starting precursors for accessing core-functionalized NDIs are bromo derivatives of naphthalenetetracarboxylic dianhydride (NDA). However, available synthetic routes require harsh reaction conditions, prolonged times, give mixtures of products, and are cost inefficient.
To overcome these problems, T. Govindaraju and co-workers, Jawaharlal Nehru Centre for Advanced Scientific Research (JNCASR), Bangalore, India, have developed 1,3-dibromo-5,5-dimethylhydantoin (DBH) as a new brominating reagent. The bromination reactions of NDA with DBH are regioselective, involve mild reaction conditions, and are cost efficient. The utility of bromo-derivatives of NDA was demonstrated by converting them into core-functionalized NDIs.
This method may be used to access other functionalized arylene diimides and aromatic systems and may make this technique attractive for industrial applications.
 Dibromohydantoin: A Convenient Brominating Reagent for 1,4,5,8-Naphthalenetetracarboxylic Dianhydride,
Dibromohydantoin: A Convenient Brominating Reagent for 1,4,5,8-Naphthalenetetracarboxylic Dianhydride,
M. Sasikumar, Y. V. Suseela, T. Govindaraju,
Asian J. Org. Chem. 2013.
DOI: 10.1002/ajoc.201300088