The design of light-activatable species involves the modification of a compound bearing an active functional group with a photocleavable moiety (protecting group) so that it remains inert until exposure to light releases the active compound. This sounds good in theory, but the downside is that many biomolecules, such as protease substrates and oligonucleotides, do not have one specific functional group that controls their activity.
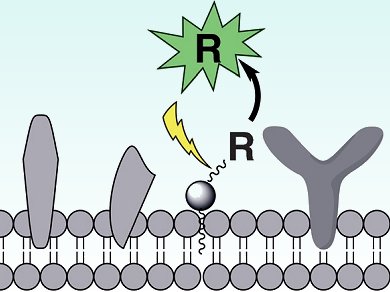
David Lawrence and co-workers, University of North Carolina, USA, have come up with a way round this problem: they attach a bioactive agent, R, and a photocleavable linker to a membrane-targeting group, in this case a lipid that can anchor to cell membranes (see picture). This way, the agent is kept hidden from the biological target in the lipid pool until the linker is cleaved upon light exposure, whereupon the active agent is released.
The researchers tested their design by embedding light-activatable species in mitochondria and erythrocytes, and were furthermore able to study intracellular proteolytic activity.
This method is thus not only useful for controlling biological processes but also has potential for drug delivery.
- Lipid Pools As Photolabile “Protecting Groups”: Design of Light- Activatable Bioagents,
Luong T. Nguyen, Nathan P. Oien, Nancy L. Allbritton, David S. Lawrence,
Angew. Chem. Int. Ed. 2013.
DOI: 10.1002/anie.201305510