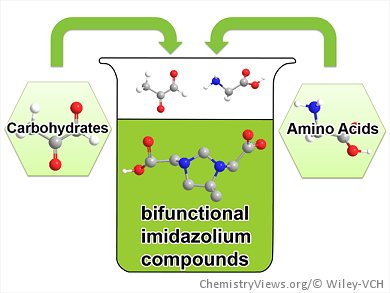
Imidazolium compounds are very versatile molecules that have enabled a large number of recent innovations. Among these, imidazolium-based ionic liquids (ILs) are the most prominent. Coming with beneficial solvent properties, negligible vapor pressure, and high thermal stability, ILs are often classified as “green solvents”. A critical analysis, however, reveals that synthesis and purification of ILs show a lot of room for improvement in terms of sustainability and cost reduction. Consequently, greener methods to access ILs are still highly welcomed and are expected to expand the range of applications of those systems.
Davide Esposito and co-workers, Max Planck Institute of Colloids and Interfaces, Potsdam, Germany, have reported a simple and efficient method for the synthesis of bifunctional imidazolium compounds based on a modification of the Debus–Radziszewski synthesis. By following the rules of green chemistry, the one-pot reactions are performed in water without excess of reagents at ambient conditions and afford a library of compounds bearing functionalities derived from natural amino acids or simple aliphatic amines in very good yields. This, together with the use of pyruvaldehyde as the 1,2-dicarbonyl compound, has resulted in a method to prepare imidazolium compounds fully based on renewable platform chemicals.
A model study on the reactivity of a representative building block was performed that showed favorable results, thus paving the way for their application in the synthesis of polymers and other complex structures.
- A Sustainable Route towards Imidazolium Building Blocks based on Biomass Molecules,
Davide Esposito, Sarah Kirchhecker, Markus Antonietti,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201302806