The Triboelectric Effect
As sources of renewable energy, sun and wind have one major disadvantage: it isn’t always sunny or windy. Waves in the ocean, on the other hand, are never still. American researchers are now aiming to use waves to produce energy by making use of contact electrification between a patterned plastic nanoarray and water. In the journal Angewandte Chemie, they have introduced an inexpensive and simple prototype of a triboelectric nanogenerator that could be used to produce energy and as a chemical or temperature sensor.
The triboelectric effect is the build up of an electric charge between two materials through contact and separation – it is commonly experienced when removal of a shirt, especially in dry air, results in crackling. Zhong Lin Wang and his team, Georgia Institute of Technology, Atlanta, USA, have previously developed a triboelectric generator based on two solids that produces enough power to charge a mobile telephone battery. However, high humidity interferes with its operation. How could this technology work with waves in water? The triboelectric effect is not limited to solids; it can also occur with liquids. The only requirement is that specific electronic energy levels of two substances are close enough together. Water just needs the right partner – maybe a suitable plastic.
Polydimethylsiloxane Pyramids
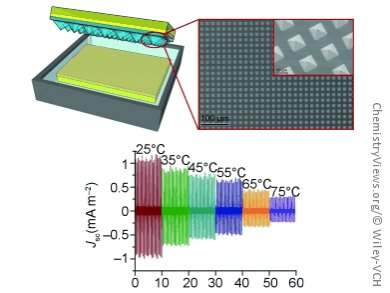
As a prototype, the researchers made an insulated plastic tank, whose lid and bottom contain copper foil electrodes. Their system is successful because the inside of the lid is coated with a layer of polydimethylsiloxane (PDMS) patterned with tiny nanoscale pyramids. The tank is filled with deionized water. When the lid is lowered so that the PDMS nanopyramids come into contact with the water, groups of atoms in the PDMS become ionized and negatively charged. A corresponding positively charged layer forms on the surface of the water. The electric charges are maintained when the PDMS layer is lifted out of the water. This produces a potential difference between the PDMS and the water. Hydrophobic PDMS was chosen in order to minimize the amount of water clinging to the surface; the pyramid shapes allow the water to drop off readily. Periodic raising and lowering of the lid while the electrodes are connected to a rectifier and capacitor produces a direct current that can be used to light an array of 60 LEDs. In tests with salt water, the generator produced a lower output, but it could in principle operate with seawater.
The current produced decreases significantly as temperature increases, which could allow this device to be used as a temperature sensor. It also decreases when ethanol is added to the water, which suggests potential use of the system as a chemical sensor. By attaching probe molecules with specific binding partners, it may be possible to design sensors for biomolecules.
- Water-Solid Surface Contact Electrification and its Use for Harvesting Liquid Wave Energy,
Zong-Hong Lin, Gang Cheng, Long Lin, Sangmin Lee, Zhong Lin Wang,
Angew. Chem. Int. Ed. 2013.
DOI: 10.1002/anie.201307249