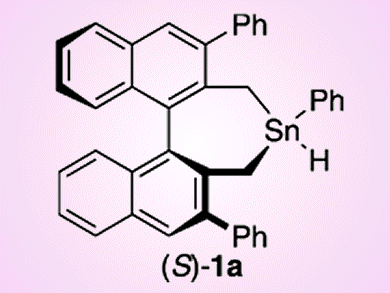
Radical cyclizations mediated by organotin hydrides are powerful tools in organic synthesis, and the development of chiral organotin-catalyzed enantioselective radical cyclization is very attractive. Although several chiral organotin hydrides have previously been prepared, an enantioselective cyclization using these chiral tin reagents has never been reported. The reason for this is that efficient transfer of chirality from the chiral organotin to the product is difficult to implement in many radical cyclizations, because the organotin reagent often only engages in atom abstraction to generate a radical in the substrate, but does not participate in the bond formation process.
Keiji Maruoka and co-workers, Kyoto University, Japan, have reported the first example of chiral organotin-hydride-catalyzed enantioselective radical cyclization of aldehydes. The radical reaction proceeds via an O-stannyl ketyl intermediate, such that chiral information from the organotin compound can be transfered to the product in the cyclization step:
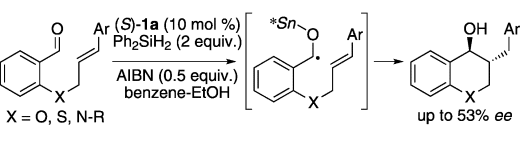
This offers an insight for the future design of catalytic asymmetric radical reactions.
- Chiral Organotin Hydride Catalyzed Enantioselective Radical Cyclization of Aldehydes,
Seiji Shirakawa, Asuka Usui, S. B. Jennifer Kan, Keiji Maruoka,
Asian J. Org. Chem. 2013.
DOI: 10.1002/ajoc.201300138