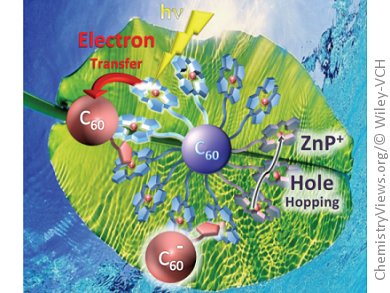
Models for natural photosynthetic systems are important in the search for an insight into the key factors that affect the primary events of these systems. Towards this end J.-F. Nierengarten, Université de Strasbourg, France, and N. Armaroli, Consiglio Nazionale delle Ricerche, Bologna, Italy, and their research groups have been investigating the photophysical properties of a supramolecular fullerene–porphyrin ensemble. This resulted from the self-assembly of a pyrrolidinofullerene–imidazole derivative (F1) with a zinc porphyrin (ZnP) array constructed around a hexasubstituted fullerene core (F(ZnP)12).
The team found that this hexasubstituted fullerene core by itself does not play any part in intercomponent photoinduced processes. However, when a monosubstituted fullerene guest F1 coordinates axially to one of the zinc porphyrins, a quantitative quenching of the fluorescence signal of the metalloporphyrin moiety was observed. In toluene, the supramolecular ensemble exhibits a charge-transfer emission centered around 930 nm, suggesting the occurrence of intramolecular face-to-face interactions of F1 with neighboring metalloporphyrin moieties within the self-assembled photoactive array.
In addition, owing to the large number of porphyrin subunits, the host system also serves as an antenna capable of harvesting the absorbed light prior to the electron-transfer event. In this way, the supramolecular ensemble resulting from the association of F(ZnP)12 and F1 mimics the primary events of natural photosynthetic systems.
- A Supramolecular Photosynthetic Model Made of a Multiporphyrinic Array Constructed around a C60 Core and a C60–Imidazole Derivative,
K. Yoosaf, J. Iehl, I. Nierengarten, M. Hmadeh, A.-M. Albrecht-Gary, J.-F. Nierengarten, N. Armaroli,
Chem. Eur. J. 2014.
DOI: 10.1002/chem.201303481