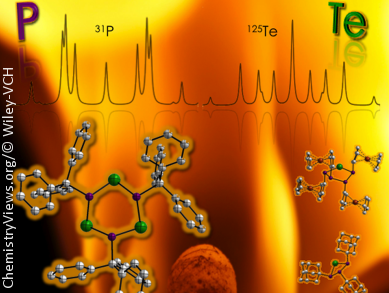
Phosphorus–tellurium compounds, in contrast to phosphorus–sulfur, which are widely used in matches, and phosphorus–selenium compounds, are quite rare because it is difficult to stabilize the P–Te and P=Te bonds. J. Derek Woollins’ group from the University of St Andrews, UK, in collaboration with groups from the Universities of Calgary, Canada, and Munich, Germany, have synthesized a series organophosphorus(III)–tellurium heterocycles by means of two new synthetic approaches.
The first involves the reaction of dichlorophosphanes RPCl2, in which R is a bulky substituent, essential for the thermal stabilization for the products, with suspended Na2Te2; precipitation of NaCl and of elemental tellurium led to the products (RP)nTem. The second pathway, which involved the introduction of a PV2N2 anchor, led to the generation of new bicylic compounds with PIII–Te–PV units. For the first time these compounds could be structurally characterized by X-ray crystallography owing to their newfound thermal stability.
- Isolatable Organophosphorus(III)–Tellurium Heterocycles,
A. Nordheider, T. Chivers, O. Schön, K. Karaghiosoff, K. S. Athukorala Arachchige, A. M. Z. Slawin, J. D. Woollins,
Chem. Eur. J. 2014.
DOI: 10.1002/chem.201303884