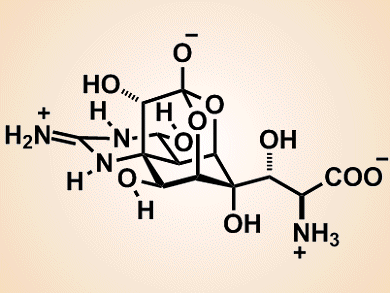
Chiriquitoxin, a potent neurotoxin, is an analogue of tetrodotoxin, in which a glycine residue is connected to the C-11 position of tetrodotoxin. Both compounds were isolated from the skin of a Costa Rican dart frog, Atelopus chiriquensis, in 1975. However, the structure of chiriquitoxin (pictured) was not elucidated until 1990. Tetrodotoxin and its analogues are ion-channel inhibitors and have, thus, attracted considerable interest in the field of neurophysiology. Chiriquitoxin is of particular interest as it is the first tetrodotoxin analogue shown to interfere with the outwardly rectifying potassium channel. However, the scarcity of chiriquitoxin from natural sources has prevented further detailed investigations into the mechanisms of the ion-channel inhibitions of these compounds. Therefore, a supply of this natural product by chemical synthesis is highly desired.
Toshio Nishikawa and co-workers, Nagoya University, Japan, have been focused on developing a subtype selective blocker of a voltage-gated sodium channel based on tetrodotoxin and saxitoxin to analyze biological functions. With chiriquitoxin being the sole example of the tetrodotoxin family that possesses potassium-channel blocking activity, this compound became a major synthetic target of the group.
With this in mind, the group has designed an intermediate for the syntheses of a wide variety of tetrodotoxin analogues, with the synthesis of chiriquitoxin achieved by a stereocontrolled aldol reaction of a D-camphor-derived iminolactone for introduction of the glycine unit and new deprotection procedure of methylthiomethyl ether by Pummerer rearrangement as the key steps. Biochemical investigations using the synthesized chiriquitoxin and its intermediate are currently underway.
- Total Synthesis of Chiriquitoxin, an Analogue of Tetrodotoxin Isolated from the Skin of a Dart Frog,
Masaatsu Adachi, Takuya Imazu, Ryo Sakakibara, Yoshiki Satake, Minoru Isobe, Toshio Nishikawa,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201304110