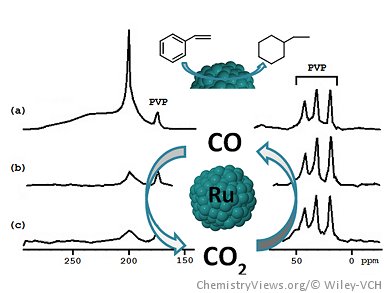
The precise location of ligands and the ability to monitor catalytic reactions is of high interest in heterogeneous catalysis. Many in situ techniques have been developed, but the use of simple alternative methods, similar to those used in molecular chemistry, for example, NMR spectroscopy, could bring additional information on the reaction sites and possibly on reaction intermediates.
Karine Philippot, Bruno Chaudret, and co-workers, Toulouse, France, have studied the surface chemistry of ruthenium nanoparticles (Ru NPs) by using GC, IR, solid-state, and gas-phase NMR spectroscopy. They have also characterized the nanoparticles by wide-angle X-ray scattering (WAXS) and X-ray photoelectron spectroscopy (XPS) analysis.
They studied three test reactions—CO oxidation, CO2 reduction, and styrene hydrogenation—and looked for evidence of the influence of the presence or absence of ancillary ligands on the reactivity of the Ru surface. Taken together their results provide a picture at the molecular level of small Ru NPs where CO binds preferably in a bridging coordination mode on sites that are also sites of arene hydrogenation, namely the face atoms. These are also the preferred sites for CO oxidation. Hydrides and terminal CO compete for sites located near the bulky diphosphine ligands and as they are inactive for arene hydrogenation, they are presumably placed at apexes and edges.
These results are a good illustration of the possibility to perform selective chemistry on the surface of nanoparticles.
- Surface Chemistry on Small Ruthenium Nanoparticles: Evidence for Site Selective Reactions and Influence of Ligands,
F. Novio, D. Monahan, Y. Coppel, G. Antorrena, P. Lecante, K. Philippot, B. Chaudret,
Chem. Eur. J. 2014, 20, 1287–1297.
DOI: 10.1002/chem.201303935