To achieve efficient ethylene/ethane gas separation with porous materials, Keisuke Kishida, Showa Denko K.K., Tokyo, Japan, Susumu Kitagawa, Kyoto University, and their co-workers investigated porous coordination polymers, also known as metal–organic frameworks, of zinc with dicarboxylate and dipyridyl ligands.
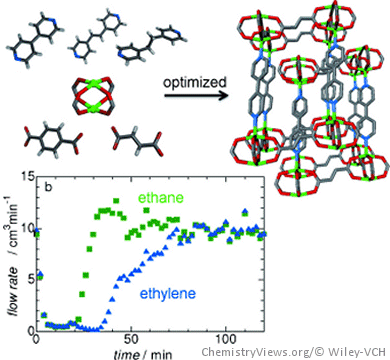
They evaluated the gas sorption properties of four pillared-layer-type porous coordination polymers with double interpenetration, [Zn2(tp)2(bpy)]n (1), [Zn2(fm)2(bpe)]n (2), [Zn2(fm)2(bpa)]n (3), and [Zn2(fm)2(bpy)]n (4) (tp = terephthalate, bpy = 4,4′-bipyridyl, fm = fumarate, bpe = 1,2-di(4-pyridyl)ethylene, and bpa = 1,2-di(4-pyridyl)ethane).
Different ligands yielded materials with different pore sizes. All compounds showed entangled, jungle-gym-like structures. The compound with optimal pore size (4) could adsorb 4.6 times more ethylene than ethane from a binary mixture of the two gases. Repeated adsorption–desorption studies revealed that the coordination polymer could be fully regenerated by gentle depressurization due its structural flexibility, while a conventional zeolite could not.
This work illustrates the advantage of flexible porous coordination polymers over traditional adsorbents in the field of gas separation, as well as a strategy of material design.
- Structural Optimization of Interpenetrated Pillared-Layer Coordination Polymers for Ethylene/Ethane Separation,
Keisuke Kishida, Satoshi Horike, Yoshihiro Watanabe, Mina Tahara, Yasutaka Inubushi, Susumu Kitagawa,
Chem. Asian J. 2014.
DOI: 10.1002/asia.201400122