Membranes with Tailored Separation Properties
Filtration using membrane filters is one of the most commonly used separation techniques. Modern developments are aimed at membranes with tailored separation properties as well as switchability. German scientists have now developed a very simple method for the modification of membranes through the inclusion of microgels. In the journal Angewandte Chemie, they introduce hollow-fiber membranes that demonstrate temperature-dependent flow and retention, thanks to thermoresponsive microgels.
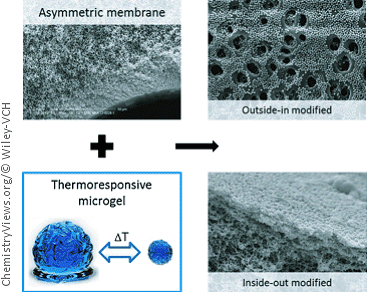
The researchers from RWTH Aachen University and DWI—Leibniz Institute for Interactive Materials, Germany, used commercially available hollow-fiber membranes used for the ultra- and microfiltration of water. Hollow-fiber filters consist of bundles of fibers made of a semipermeable material with a channel on the inside. The walls of the fibers act as the membrane. In order to modify the membranes, a team led by Matthias Wessling simply filtered microgel suspensions through them. This allowed the microgels to become embedded into the porous structure of the membrane.
A gel is a three-dimensional molecular network that is filled with a liquid. Unlike the liquid in a sponge, the liquid in the gel is tightly bound. Microparticles of a gel are called microgels. The researchers used microgels made of polyvinylcaprolactam that are stable to about 32 °C. Above this temperature, the gel structure collapses, letting the water out.
Hollow-Fiber Membranes Loaded with Microgels
The hollow-fiber membranes used have an asymmetric pore structure with internal diameters of 30 nm (ultrafiltration) to 200 nm (microfiltration) and external diameters of several micrometers. The fibers can be loaded with the microgels either from the outside in or from the inside out. In the first method the gel particles penetrate partway into the pores and the outside is then coated with microgel. In the second method, gel particles are only found on the interior of the pores, but not in those pores that are very narrow. In both types of fibers, the microgel is so firmly adsorbed that it cannot be washed away either during filtration or back flushing.
The permeability of both types is significantly reduced relative to the untreated membrane, because the microgel makes the pores less accessible. Raising the temperature causes the microgel to shrink, increasing the permeability of the membrane; cooling reverses the effect. This switching mechanism could be an important method for an efficient cleaning of the hollow fiber when high flow rates are needed during a backwashing step at low temperatures.
“The modification of conventional hollow-fiber membranes with stimuli-responsive microgels provides a straightforward and versatile route to design functional membranes with new, tailored properties that allow for regulation of the permeability,” says Wessling. “Varying the chemical structures of the microgels allows for the introduction of specific functionalities into membranes, increasing the efficiency and selectivity of separation processes in water treatment and medical technology. We will further develop this versatile platform by fundamental research within the SFB Functional Microgels and Microgel Systems of the German Research Foundation (DFG).”
- Temperature-Modulated Water Filtration Using Microgel-Functionalized Hollow-Fiber Membranes,
Daniel Menne, Fee Pitsch, John E. Wong, Andrij Pich, Matthias Wessling,
Angew. Chem. Int. Ed. 2014.
DOI: 10.1002/anie.201400316