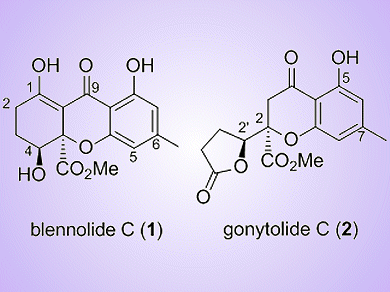
Tetrahydroxanthenone natural products are of increasing interest owing to their intriguing structural features and wide range of biological activities, for example, antimicrobial, antibiotic and cytotoxic activities. This class of natural products includes compounds such as blennolide C (1), isolated from the endophytic fungus Blenoria sp., and gonytolide C (2), isolated from the fungus Gonytrichum sp.
Lutz F. Tietze, Georg-August-University, Göttingen, Germany, and co-workers have developed the first enantioselective total syntheses of (−)-blennolide C and (−)-gonytolide C, as well as some related chromanones. The key step of these syntheses is an enantioselective Pd-catalyzed domino-Wacker/carbonylation/methoxylation reaction, combined with a diastereoselective dihydroxylation reaction. The natural products were formed with ee values of 99 %.
The researchers have successfully applied a versatile domino process to the enantioselective synthesis of a rapidly growing class of natural products.
- A Domino Approach to the Enantioselective Total Syntheses of Blennolide C and Gonytolide C,
Lutz F. Tietze, Stefan Jackenkroll, Judith Hierold, Ling Ma, Bernd Waldecker,
Chem. Eur. J. 2014.
DOI: 10.1002/chem.201402495