Parallel Solid-phase Synthesis
The synthesis of peptides and proteins is an extremely complex matter, because they must be built up one amino acid at a time in repeated reaction and washing steps. Solid-phase synthesis has made this easier. Canadian researchers introduced a new variant of this technique in the journal Angewandte Chemie. In their system, parallel syntheses take place in small regions on a paper support. The resulting peptide arrangements can be used in cell-based assays or in the search for 3D materials that support cell adhesion and growth.
In classical solid-phase synthesis, the growing peptide chain is fixed to a solid support—usually a polymer bead—so that the reagents can be rapidly and easily washed away after each step. Parallel solid-phase synthesis, known as SPOT synthesis, was developed as an alternative in the 1990s. This allows a large number of peptides to be obtained on a planar support with a small surface area. SPOT synthesis has since been adapted for other applications, such as cell-based screening. The problem is that existing SPOT systems are not well-suited for chemical reactions. When individual drops of reagent are added by pipette, they wet small areas of the membrane—the SPOTs. The circular spot of solvent absorbed by the membrane determines the size of the “reaction vessel”. Unlike in classical solid-phase synthesis, this limits the amounts of reagent, and flow-through conditions are not possible. This significantly limits the possible yields of the reactions.
New Synthesis of Peptides and Proteins
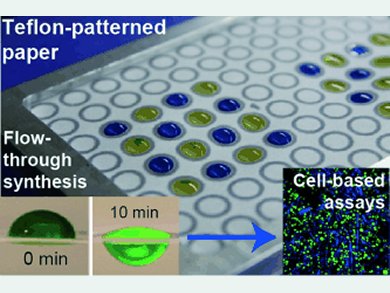
A team headed by Frédérique Deiss and Ratmir Derda at the University of Alberta, Canada, has found an elegant solution to this problem. The researchers used a Teflon coating to form a pattern of solvent-repellent barriers on a paper support. The pattern restricts the liquids to specific Teflon-free zones on the paper, forming small “reaction vessels” that can hold a larger volume than the usual SPOTs. This not only allows for the use of excess volumes of reagents, but also allows for a flow-through reaction because the larger volume ensures for gravity-driven flow of the reagent solution through the paper. The flow rate can be varied by using paper of different porosity. This significantly improves yields.
There is an additional advantage to this method: the paper can be stacked or folded into thicker three-dimensional structures. The researchers were able to identify various peptides among those immobilized on the surface that support cell adhesion, growth, or differentiation in a three-dimensional environment.
- Flow-Through Synthesis on Teflon-Patterned Paper to Produce Peptide Arrays That Can Be Used for Cell-Based Assays,
Frédérique Deiss, Wadim L. Matochko, Natasha Govindasamy, Edith Y. Lin, Ratmir Derda,
Angew. Chem. Int. Ed. 2014.
DOI: 10.1002/anie.201402037