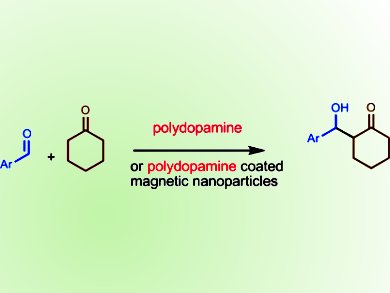
Polydopamine (PDA) is a polymer formed by oxidation of dopamine. Although its structure is still under discussion, there is a consensus that it is built up of indole units of different states of hydrogenation, mainly connected by C–C bonds between the benzene rings. PDA coatings have found various biomedical applications, such as in drug transport and delivery, as antibacterial coatings, in separation of biological materials or poisonous contaminants, and as supports for organocatalytic moieties. PDA is generally considered to be inert and thus nonpoisonous and biocompatible in biomedicine.
Jürgen Liebscher, Humboldt-University Berlin, Germany, and and colleagues from there and the National Institute of Research and Development for Isotopic and Molecular Technologies, Cluj-Napoca, Romania, have conducted a study that may now change the view that PDA is just an innocent inert polymer. The discovery was made by the group whilst they were conducting research on the use of proline-containing PDA-covered magnetite nanoparticles (MNP) as organocatalysts in direct aldol reactions. Instead of obtaining the enantiomerically pure aldol product, a racemate was obtained. Further investigation revealed that a nonstereoselective background reaction was being catalyzed by the PDA coating.
This discovery serves as a warning to scientists that PDA is not just an innocent polymer and may cause unwanted side effects in important applications, such as in biomedicine or catalysis.
- Polydopamine—An Organocatalyst Rather than an Innocent Polymer,
Radosław Mrówczyński, Alexander Bunge, Jürgen Liebscher,
Chem. Eur. J. 2014.
DOI: 10.1002/chem.201402532