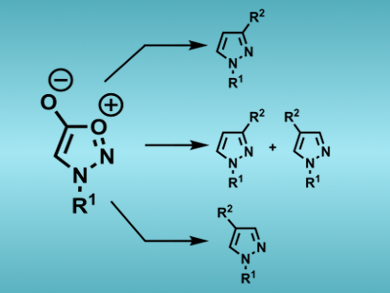
Sydnones are mesoionic heteroaromatic compounds which undergo a wide range of functionalization reactions. From a synthesis standpoint, the cycloaddition reaction of these compounds with alkynes offers an effective method for the preparation of pyrazoles. A current limitation of sydnone cycloadditions is the requirement to conduct these reactions at elevated temperatures over extended time periods. Nonetheless, such processes can provide the corresponding pyrazoles with excellent levels of regiocontrol, especially for terminal alkyne substrates.
Joseph Harrity and colleagues, University of Sheffield, UK, and Universidad del País Vasco, Spain, have found that copper salts can be used to promote the cycloaddition reaction of sydnones and terminal alkynes providing a significant reduction in reaction times. Upon the use of Cu(OTf)2, it was found that 1,3-disubstituted pyrazoles could be synthesized, whereas upon switching to Cu(OAc)2, the corresponding 1,4-isomers were produced. The mechanism of the Cu-effect in each case was investigated by experimental and theoretical studies, which suggest that Cu(OTf)2 functions by Lewis acid activation of the sydnone whereas Cu(OAc)2 promotes the formation of reactive CuI acetylides.
- Cu-Promoted Sydnone Cycloadditions of Alkynes: Scope and Mechanism Studies,
Júlia Comas-Barceló, Robert S. Foster, Béla Fiser, Enrique Gomez-Bengoa, Joseph P. A. Harrity,
Chem. Eur. J. 2015.
DOI: 10.1002/chem.201406118