Selective carbon-carbon bond formation to aromatic and heteroaromatic systems is an important synthetic objective, as the resulting structural motifs are found in many pharmaceuticals, agrochemicals and functional materials. Such transformations are currently achieved using late transition-metal mediated couplings. However, issues with the cost and future supply of such metals and the metal contamination of products makes the development of metal-free coupling processes of great importance.
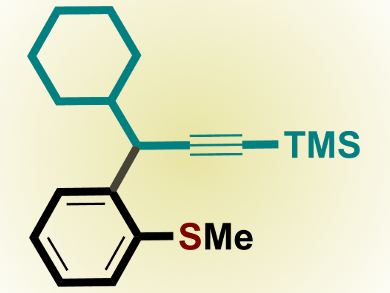
Currently, in the propargylation of aromatic and heteroaromatic systems, many coupling methods rely on electrophilic Friedel-Crafts-type processes that require stoichiometric metal reagents. David J. Procter and co-workers, University of Manchester, UK, have overcome this problem by the development of a sulfoxide-directed ortho-propargylation of aromatics and heteroaromatics that proceeds by a new interrupted Pummerer-allenyl thio-Claisen rearrangement sequence involving allenyl sulfonium salts.
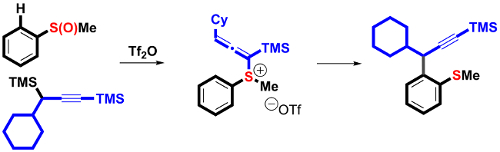
The operationally simple, metal-free procedure is general, regiospecific with regard to the propargyl nucleophile, and shows complete selectivity for products of propargylation over allenylation. In addition, the organosulfanyl group and the alkyne motif in the coupling products are versatile handles for further manipulation.
- Sulfoxide-Directed Metal-Free ortho-Propargylation of Aromatics and Heteroaromatics,
Andrew J. Eberhart, Harry J. Shrives, Estela Alvarez, Amandine Carrër, Yuntong Zhang, and David J. Procter*
Chem. Eur. J. 2015.
DOI: 10.1002/chem.201406424