Researchers from Heriot-Watt University, Edinburgh have combined boron and organometallic chemistry, catalysis, electron transfer, and DFT calculations in a study that describes the preparation and characterization of a “fly-over bridge” species.
Stuart Macgregor, Alan Welch, and co-workers report a low-temperature metal insertion into a highly stable arene carbon-carbon bond. Reduction of 1,1’-bis(o-carborane) and subsequent 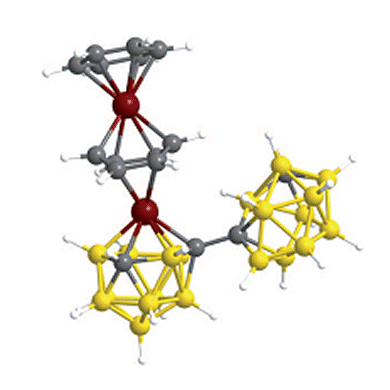 reaction with a source of {Ru(p-cymene)}2+ fragments yields a species resulting from cleavage of a p-cymene ligand. According to DFT calculations, to achieve the metal insertion a second borane cluster of the metallacarborane species is necessary, which by intramoleuclar electron-transfer reductively cleaves the aromatic moiety and leads to bond breakage.
reaction with a source of {Ru(p-cymene)}2+ fragments yields a species resulting from cleavage of a p-cymene ligand. According to DFT calculations, to achieve the metal insertion a second borane cluster of the metallacarborane species is necessary, which by intramoleuclar electron-transfer reductively cleaves the aromatic moiety and leads to bond breakage.
- Room-Temperature C-C Bond Cleavage of an Arene by a Metallacarborane
D. Ellis, D. McKay, S. A. Macgregor, G. M. Rosair, A. J. Welch
Angew. Chem. Int. Ed. 2010, 49.
DOI: 10.1002/anie.201001555 - D. Ellis, D. McKay, S. A. Macgregor, G. M. Rosair, A. J. Welch
Angew. Chem. 2010, 122.
DOI: 10.1002/ange.201001555