The enantioselective synthesis of 1-substituted tetrahydroisoquinoline derivatives has been subject to much study as it represents a versatile route to a wide range of biologically and pharmacologically active natural products.
Now, Mercedes Amat and colleagues, University of Barcelona, Spain, report a simple synthesis of an oxazolopiperidone lactam scaffold and a general procedure for the stereocontrolled introduction of substituents at the 1-position in the European Journal of Organic Chemistry.
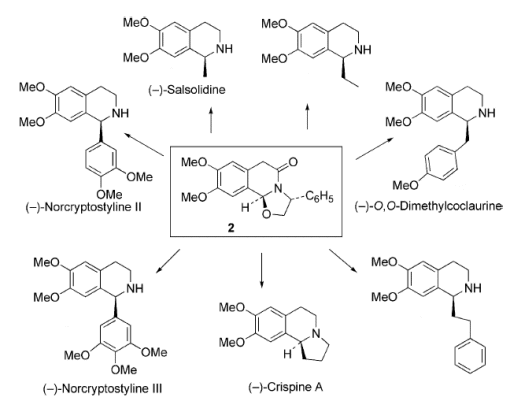
This methodology allowed the total synthesis of several alkaloids including,
- (-)-salsolidine,
- (-)-norcryptostyline II and (-)-norcryptosyline III,
- (-)-O,O-dimethylcoclauurine, and
- (S)-(-)-crispine A
along with the formal synthesis of others.
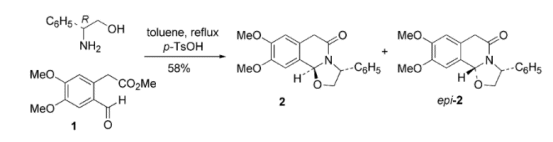
The approach involved cyclocondensation of aldehyde ester 1 with (R)-phenylglycinol to form the tricyclic lactam 2 which was the pivotal intermediate, with only small amounts of the epimeric version, epi–2, being formed.
The team then showed that Grignard reagents could be used in THF at 5 ºC to introduce substituents at the 1-position. The Grignards used ranged from methylmagnesium chloride, leading to the synthesis of (-)-salsolidine, to 2-(1,3-dioxan-2-yl)ethylmagnesium bromide, leading to the synthesis of crispine A.
Amat explains the retention of configuration at the methine carbon through coordination of the Grignard reagent with the oxygen of the oxazolidine ring. This delivers the alkyl group to the same face, preserving the stereochemistry.
As Amat notes, the scaffold approach to this enantioselective synthesis is of interest, not only because it provides general access to 1-substituted tetrahydroisoquinolines, but because it opens up a route to more complex alkaloids bearing the tetrahydroisoquinoline moiety.
- A General Methodology for the Enantioselective Synthesis of 1-Substituted Tetrahydroisoquinoline Alkaloids
M. Amat, V. Elias, N. Llor, F. Subrizi, E. Molins, J. Bosch
Eur. J. Org. Chem. 2010, 2010.
DOI: 10.1002/ejoc.201000473




