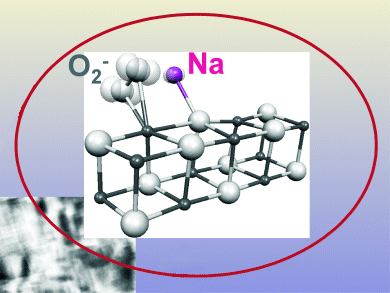
The chemistry of oxygen species at solid surfaces has attracted attention over the years owing to its relevance in fields of modern science such as heterogeneous catalysis, material science, electrochemistry, and corrosion. The bonding of molecular oxygen to metal atoms involving the formation of O2– ions is thus particularly important as it represents the initial stage of the long process that leads to the incorporation of oxygen into oxide lattices.
M. Chiesa et al. from the Università degli Studi di Torino, Italy, have tracked down the reaction pathway between sodium atoms supported at the surface of magnesium oxide and oxygen molecules by using a combined electron paramagnetic resonance (EPR) spectroscopy and quantum chemical approach. Na atoms deposited on polycrystalline MgO samples are brought into contact with O2, which are then reduced to superoxide species that are anchored at different surface sites.
The bonding of molecular oxygen to metal atoms that leads to the formation of O2– ions represents the first step in the oxygen reductive activation, and the initial stage of a long process that leads to the incorporation of oxygen into oxide lattices.
- Formation of Superoxo Species by Interaction of O2 with Na Atoms Deposited on MgO Powders: A Combined Continuous-Wave EPR (CW-EPR), Hyperfine Sublevel Correlation (HYSCORE) and DFT Study
F. Napoli, M. Chiesa, E. Giamello, G. Preda,C. Di Valentin, G. Pacchioni,
Chem. Eur. J. 2010, 16 (23).
DOI: 10.1002/chem.201000546