Lithium ion batteries (LIBs) are commonly used for electrochemical energy storage, for example in portable electronic devices and electric vehicles. However, increasing demand for lithium and depleting reserves make it necessary to look for cost-effective and sustainable alternatives. Sodium ion batteries (NIBs) are promising alternatives. Sodium, while providing lower energy densities than lithium, has a suitable redox potential for use in batteries and is an abundant element. Still, new materials and additional insight are needed to make NIBs a viable alternative for ambient-temperature energy storage.
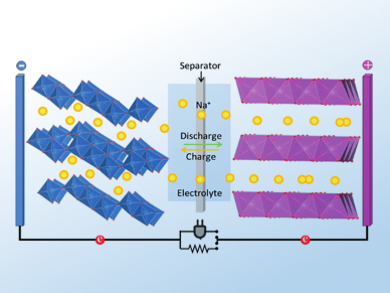
Linda F. Nazar and colleagues, University of Waterloo, Ontario, Canada, give an overview of recent developments concerning electrode materials and electrolytes for sodium ion batteries. Analogous to LIBs, the cathode materials need to intercalate sodium ions, which are transported through the electrolyte during charging (pictured), while sodium atoms are stored in the anode material.
There are several good options for cathode materials, e.g. layered sodium transition-metal oxides. In contrast, finding suitable anode materials is a bigger challenge: Elemental sodium is unsuitable for commercial applications because of safety concerns, and in contrast to lithium, sodium atoms do not intercalate in graphitic carbons. There have, however, been developments in using disordered carbons, low-potential transition metal oxides, and sodium alloys which are promising steps on the way to viable batteries.
Overcoming the multitude of challenges facing sodium ion batteries could allow competitive, cheap, sustainable energy storage for bulk and stationary purposes.
- The Emerging Chemistry of Sodium Ion Batteries for Electrochemical Energy Storage,
Dipan Kundu, Elahe Talaie, Victor Duffort, Linda F. Nazar,
Angew. Chem. Int. Ed. 2015.
DOI: 10.1002/anie.201410376
BASF celebrates its 150th anniversary this year. As part of the celebrations, Angewandte Chemie publishes a special jubilee issue where this article is included.
Also of Interest
- More articles related to the 150th anniversary of BASF
- Linda Nazar: Developing Materials for Energy Storage And Conversion (Video),
ChemViews Mag. 2013.
DOI: 10.1002/chemv.201300104 - We Try Not To Call Them Batteries,
C. Remenyi,
Nachr. Chem. 2013, 1223–1224.
DOI: 10.1002/nadc.201390404