Performance of Lithium Ion Batteries
How can the electrodes of batteries be made more efficient? In the journal Angewandte Chemie, American scientists describe a powerful approach that uses solvated graphene frameworks as the anode material. Assembled in a lithium coin cell, the as-made electrode excelled with capacities surpassing the values of typically used graphite.
The performance of lithium ion batteries relies on the properties of the electrode materials. In particular, the material not only has to be highly conductive, it also has to provide the excellent porosity needed for quick insertion and re-insertion of lithium atoms during the charge–discharge cycles. The most commercially popular anode material is graphitic carbon. However, as the authors outline, this material “cannot meet the increasing requirements for next-generation lithium ion batteries” because of “its low theoretical capacity and poor rate capability.”
From Graphene Hydrogels to High-performance Anodes
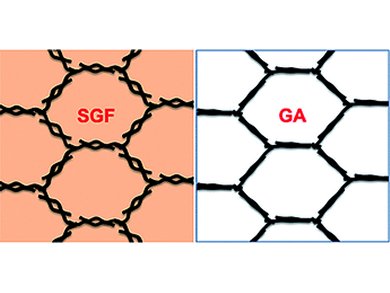
Graphene, on the other hand, is only one layer of carbon thick and, theoretically, offers a larger surface area and excellent mechanical flexibility. However, practically, it is difficult to process graphene into the bulk LIBs without aggregation, that is, severe reduction in surface area. In this context, Xiangfeng Duan and his team at the University of California, Los Angeles, USA, report on the preparation of a graphene hydrogel, which can be easily converted into solvated graphene frameworks. These frameworks are stable 3D porous structures offering both fast lithium exchange and high conductivity.
To prepare the graphene frameworks, the scientists used a modified hydrothermal method to generate free-standing cubes of a graphene hydrogel from graphite oxide. Simple solvent exchange then converted the hydrogel structures into the 3D solvated graphene frameworks, which could be readily pressed in films needed for LIB coin cells without losing their porous graphene network. These as-made anodes thus not only provided for much faster lithium diffusion, but also retained the large surface area and excellent conductivity of graphene sheets.
In addition, Duan and his team found superior electrochemical performances of their solvated graphene frameworks to that of graphene aerogels, which have also been investigated intensively for their use as replacements for the graphite electrodes. Finally, the scientists emphasize that their research also has a more general impact on battery technology as the “fundamental insights” will “promote the rational design and synthesis of 3D graphene materials for electrochemical energy storage and
- Solvated Graphene Frameworks as High-Performance Anodes for Lithium-Ion Batteries,
Yuxi Xu, Zhaoyang Lin, Xing Zhong, Ben Papandrea, Yu Huang, Xiangfeng Duan,
Angew. Chem. Int. Ed. 2015.
DOI: 10.1002/anie.201500677