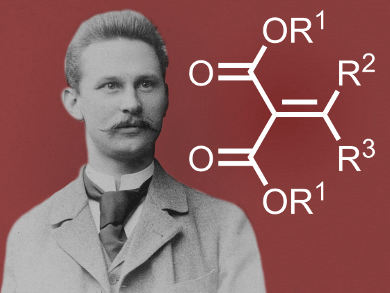
Emil Knoevenagel, inventor of the eponymous aldol condensation variant, was born on June 18, 1865, in Linden, Germany. He studied chemistry at the Technical University of Hanover and at the University of Göttigen, both Germany. His teachers included Hermann Ost, Wilhelm Kohlrausch, and Ludwig Gattermann. Knoevenagel received his Ph.D. in organic chemistry in 1889 under the supervision of Victor Meyer in Göttingen. He then joined the University of Heidelberg, were he remained until his death on August 11, 1921. He served as a field officer in World War I from 1914 to 1918.
Knoevenagel focused predominantly on organic synthesis, especially reactions of carbonyl compounds. He developed an aldol reaction leading to unsaturated products under base catalysis, named “Knoevenagel condensation” after him. He also worked on stereochemistry and heterocycle synthesis.
The Knoevenagel reaction, also known as Knoevenagel condensation, is an important reaction in organic chemistry. It is a special type of Aldol reaction that involves the reaction of ketones (R2 = organic group, R3 = organic group) or aldehydes (R2 = H, R3 = H or organic group) with compounds that have CH-acidity (R1= organic group), such as malonic ester, acetoacetic ester, or nitromethane. This reaction results in the formation of unsaturated condensation products.
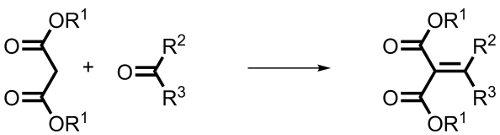
A base is added as a catalyst.The addition of the base leads to the deprotonation of the CH-acidic compound in the first step. This generates an anion, known as a carbanion, which acts as a nucleophile and attacks the carbonyl carbon of the carbonyl compound. Simultaneously, the oxygen of the carbonyl group gets protonated. As a result, an alcohol is formed. The alcohol can be isolated as an intermediate. Finally, through the elimination of water, the unsaturated end product is produced.
References
- Emil Knoevenagel (Obituary, in German),
Z. Angew. Chem. 1922, 35, 29–30.
DOI: 10.1002/ange.19220350503
Selected Publications by Emil Knoevenagel
- Ueber Condensationsproducte von Acetylaceton mit Aldehyden (in German),
E. Knoevenagel,
Ber. Dtsch. Chem. Ges. 1903, 36, 2136–2180.
DOI: 10.1002/cber.190303602134 - Condensation von Malonsäure mit aromatischen Aldehyden durch Ammoniak und Amine (in German),
E. Knoevenagel,
Ber. Dtsch. Chem. Ges. 1898, 31, 2596–2619.
DOI: 10.1002/cber.18980310308
Emil Knoevenagel is the answer to Guess the Chemist (42).
Also of Interest
- Emil Knoevenagel and the Roots of Aminocatalysis,
Benjamin List,
Angew. Chem. Int. Ed. 2010, 49, 1730–1734.
DOI: 10.1002/anie.200906900