1D and 2D highly π-conjugated aromatic molecules are of great interest as potential organic semiconductors. Among these, polycondensed aromatic hydrocarbons with reduced symmetries are garnering increasing attention. Derivatives based on the large aromatic core tribenzo[fj,ij,rst]pentaphene (TBP) are advantageous over other polycondensed aromatic hydrocarbons because of their versatile synthesis and the easy access to many possible functionalization sites.
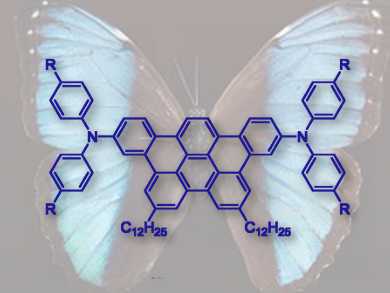
Titus A. Jenny, University of Fribourg, Switzerland, and colleagues have synthesized two tribenzo[fj,ij,rst]pentaphene (TBP) derivatives, 3,12-bis(diphenylamino)-6,9-didodecyltribenzo[fg,ij,rst]pentaphene and 3,12-bis[di(4’-octylphenyl)amino)]-6,9-didodecyltribenzo[fg,ij,rst]pentaphene. The derivatives bear two diarylamine substituents attached at the opposite ends of the aromatic core.
Field effect transistor (FET) devices incorporating the TBP compounds were made using spin coating under different concentrations, spin speed, and solvent conditions. Emission spectra and surface investigation by atomic force microscopy (AFM) showed that aggregates formed caused by the strong π–π stacking of the aromatic core. This lead to island features, and thus, to unexpectedly low hole mobilities.
According to the researchers, their synthetic strategy offers the possibility to decorate the TBP core structure with various charge-carrier peripheral groups and optimized alkyl chains. This should improve the crystalline properties of their thin films upon deposition and lead to a better hole transport mobility.
- Synthesis of Arylamine Tribenzopentaphenes and Investigation of their Hole Mobility,
Bassam Alameddine, Andrew H. Rice, Christine Luscombe, Titus A. Jenny,
ChemistryOpen 2015.
DOI: 10.1002/open.201500064