Iodine-capture materials have important applications in the management of byproducts generated from nuclear fission of uranium, as 129I has a long radioactive half-life (1.57·107 years) and can be incorporated into the human metabolism. Current techniques for iodine capture using natural or synthetic zeolites suffer from limited adsorption capacity.
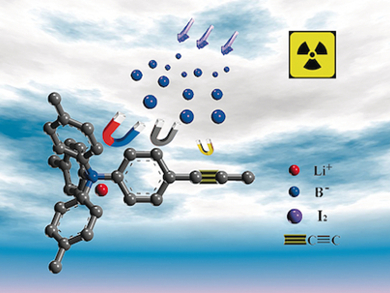
Guangshan Zhu and colleagues, Northeast Normal University, Changchun, China, investigated the use of porous aromatic frameworks (PAFs) for the efficient capture of iodine. PAF-23, PAF-24, and PAF-25 were synthesized from tetrahedral borate building blocks and various alkyne monomers. The presence of three effective sorption sites – the ionic bond, the phenyl ring, and the triple bond – led to high affinity and capacity for iodine. The PAFs also exhibited excellent thermal and chemical stabilities.
Gravimetric iodine-uptake analysis revealed the highest value of 2.76 g g−1 for PAF-24, which represents the highest sorption value for all porous materials (zeolites, MOFs, and POFs). The nature of this bonding was further explored using additional PAF analogues, confirming the role of the three sorption sites in efficient iodine capture.
- Highly Efficient Enrichment of Volatile Iodine by Charged Porous Aromatic Frameworks with Three Sorption Sites,
Zhuojun Yan, Ye Yuan, Yuyang Tian, Daming Zhang, Guangshan Zhu,
Angew. Chem. Int. Ed. 2015.
DOI: 10.1002/anie.201503362
|
The porous aromatic framework PAF-24 was synthesized from lithium tetrakis(4-iodophenyl)borate and an alkyne monomer and reported to exhibit the highest iodine adsorption capacity for any porous material (2.76 g g−1). |
 Find more world records from all branches of chemistry on the Records and Challenges platform of
Find more world records from all branches of chemistry on the Records and Challenges platform of 


