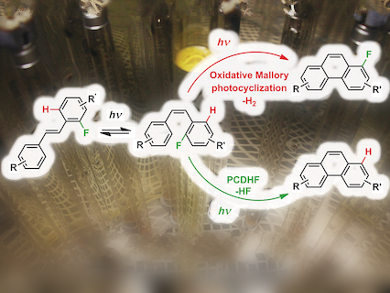
Zhe Li and Robert Twieg, Kent State University, OH, USA, have reported that the Mallory reaction of o-terphenyls with fluorinated terminal rings is a facile and general route to fluorinated polyaromatics. The reaction proceeds via an elimination mechanism with loss of HF.
The synthesis of fluorinated polyaromatic hydrocarbons is of great interest for their applications in medicine and materials science. Photochemical conversion of stilbenes into phenanthrene derivatives has been known since the 1930s and the reaction, known as the Mallory reaction, is also applicable to the formation of heterocyclic polyaromatics. The reaction tends to require external oxidants, such as iodine, Ph2Se2, or copper salts.
The Mallory reaction has been known to proceed via an elimination mechanism, whereby photocyclization brings about loss of HX (X is a functional group on stilbene). However, fluorine has rarely been regarded as a potential X group for eliminative photocyclization.
Li and Twieg found the photocyclodehydrofluorination reaction proceeded in the absence of an oxidant in just as facile a manner as other photocyclodehydrohalogenation reactions and was also applicable to heterocyclic polyaromatics.
- Photocyclodehydrofluorination,
Zhe Li, Robert J. Twieg,
Chem. Eur. J. 2015.
DOI: 10.1002/chem.201502473