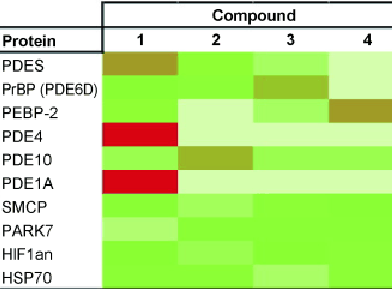
The difficult task of determining off-target effects during drug development has become much easier thanks to research from a collaborative effort led by Albert J. R. Heck, University of Utrecht, The Netherlands. Heck’s team used a chemical proteomics approach to enrich for proteins that bind to analogues of known phosphodiesterase 5 inhibitors. They assessed the binding of a large set of proteins to a small library of inhibitors and thus determined individual selectivity profiles for these compounds.
Their results show that even minor chemical modifications to these compounds can significantly bias their selectivity toward other interacting proteins. This approach could become a valuable tool for designing out undesired off-target interactions in vivo and for discovering new targets of established drug molecules.
- Target Profiling of a Small Library of Phosphodiesterase 5 (PDE5) Inhibitors using Chemical Proteomics
R. Raijmakers, P. Dadvar, S. Pelletier, J. Gouw, K. Rumpel, A. J. R. Heck,
ChemMedChem 2010, 5(11).
DOI: 10.1002/cmdc.201000303