Tunable Photoswitches
A modular approach to molecular photoswitches has been devised by chemists in Germany and allowed them to build a library of some 40 stable acylhydrazones. They have demonstrated how widely tunable these stable photoswitches are and point to a potential range of applications in biology, supramolecular chemistry as well as materials science, where these compounds enable light control over molecular systems and their properties.
Chemists Derk Jan van Dijken, Petr Kovaříček, and Svante Ihrig in the team of Stefan Hecht at the Humboldt University of Berlin, Germany, remark in the Journal of the American Chemical Society how molecular photoswitches have attracted considerable interest from several disparate fields in science. There are many well-known and well-studied examples, including those based on stilbenes and azobenzenes, diarylethenes and spiropyrans.
But, as the researchers state, “distinct obstacles and inherent limitations remain” to their further development and few novel photoswitches have emerged in recent decades. An important issue facing chemists hoping to develop these compounds is the significant synthetic effort required to construct such functional compounds, as well as the complex link between molecular structure and the associated switching function. “The rational design of photoswitches is typically challenging and not straightforward, resulting in lengthy syntheses of complicated molecules whose desired properties cannot be probed until the final stage,” the team points out.
Acylhydrazones as Flexible Building Blocks
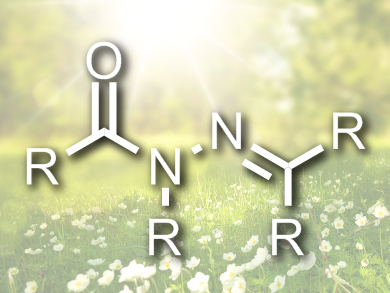
Inspired by work from Jean-Marie Lehn [1] and Ivan Aprahamian [2], Hecht and colleagues have turned to acylhydrazones, with their imine structural motif. These compounds have great promise in this area but have not been the focus of much effort previously. The backbone of these molecules has four positions for functionalization, at each of which a different substituent can be introduced to allow the tuning of switching behavior.
The researchers have synthesized over three dozen variations on the acylhydrazone theme that can be constructed in a modular manner in an easy atom-economic synthesis from readily available starting materials. They have also demonstrated that the compounds are robust and have “superb ambient stability” and fatigue resistance.
A Wide Range of Possible Functionalities
In the design of novel photoswitches, however, it is their functionality, and most importantly their response to light that is paramount. There are four performance parameters – addressability, thermal stability, efficiency, and reliability – that define a useful photoswitch. The acylhydrazones display good quantum efficiency, with high quantum yields and photostationary states. They are “addressable” with good band separation, molar absorbtivity and useful absorption maxima.
Indeed, “by proper substitution,” the researchers report, they can achieve, “excellent band separation between the absorbance maxima of the (E)- and (Z)-isomers in the UV or visible region.” The prepared switches span a wide range of thermal half-lives, from short-lived T-type all the way to thermally stable P-type derivatives. In addition, the team also found a rare negative photochromic system.
The researchers suggest that they have unraveled the link between structure and activity for photoswitches. This, the facile preparation, and the chemical robustness of precursors and products opens up the possibility of stable designer photoswitches with on-demand properties. “One particular advantage lies in the reversibility of the central C=N bond formation, which is unique among photoswitches and [we] are now trying to explore,” Hecht told ChemViews Magazine. “We are actively trying to integrate this new class of photoswitches into our design of remote-controlled catalytic and (supra)molecular systems”, the team concludes.
“The acylhydrazone switches are an exciting addition to the ever-expanding family of photochromic compounds,” says Ivan Aprahamian of Dartmouth College, Hanover, NH, USA. “Their ease of synthesis and controllable lifetimes will likely expedite their deployment in different applications, while their excellent band separation will lead to efficient switching processes.”
References
- [1] Configurational and Constitutional Information Storage: Multiple Dynamics in Systems Based on Pyridyl and Acyl Hydrazones,
Manuel N. Chaur, Daniel Collado, Jean-Marie Lehn,
Chem. Eur. J. 2011, 17, 248–258.
DOI: 10.1002/chem.201002308 - [2] A switching cascade of hydrazone-based rotary switches through coordination-coupled proton relays,
Debdas Ray, Justin T. Foy, Russell P. Hughes, Ivan Aprahamian,
Nat. Chem. 2012, 4, 757–762.
DOI: 10.1038/nchem.1408
- Acylhydrazones as Widely Tunable Photoswitches,
Derk Jan van Dijken, Petr Kovaříček, Svante P. Ihrig, Stefan Hecht,
J. Am. Chem. Soc. 2015.
DOI: 10.1021/jacs.5b09519