Mimicry of the [FeFe]-hydrogenase active site has drawn much attention with a view to generating models for renewable energy storage because hydrogenase enzymes reversibly catalyze hydrogen activation.
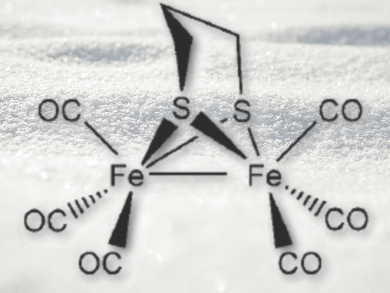
To study the photochemical reactivity of the [FeFe]-hydrogenase active-site model [(μ-pdt){Fe(CO)3}2] (pdt=1,3-propanedithiolate) and analogous complexes with different dithiolate bridging ligands (pictured), Wyatt Thornley and Thomas Bitterwolf, University of Idaho, Moscow, USA, performed photolysis of the model complexes in low-temperature frozen matrices.
Through characterization by FTIR spectroscopy and DFT calculations, they were able to identify a far wider range of photolysis products of the model complexes than were observed in previous ultrafast solution studies. This is due to the increased stability of photogenerated species at cryogenic temperatures, allowing for multiphoton reactions. Besides the previously identified basal decarbonylation products, apical carbonyl-loss isomers were also identified, as were products of β-hydride activation of the alkyldithiolate bridge via secondary photolysis, and, from the benzenedithiolate-bridged complex, formation of a 10 π-electron aromatic metallacycle.
- Intramolecular C–H Activation and Metallacycle Aromaticity in the Photochemistry of [FeFe]-Hydrogenase Model Compounds in Low-Temperature Frozen Matrices,
Wyatt A. Thornley, Thomas E. Bitterwolf,
Chem. Eur. J. 2015.
DOI: 10.1002/chem.201503826