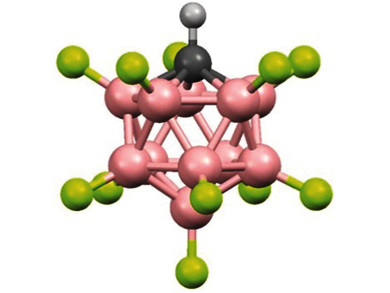
Very strong acids offer the intriguing possibility of protonating molecules that have not previously been protonated in the lab. However, the ranking of acid strength for these acids is not trivial. The proton-bridged polymeric structures of solid carborane acids hamper their reaction with gases due to slow kinetics at the solid–gas interface. Conversely, dissolving such acids in a solvent inherently means leveling of their acidity down to that of the protonated solvent.
To determine whether the carborane acid H(CHB11F11) or traditional mixed Lewis/Brønsted superacids hold claim to the title of strongest acid, Christopher Reed and colleagues at the University of California, USA, took the pragmatic approach of observing which of these acids were capable of successfully protonating the weakest base.
Exposure of H(CHB11F11) to CO2 led to the formation of a protonated CO2 species, as confirmed by IR spectroscopy. This CO2 could be displaced by the addition of a stronger base (benzene!). DFT calculations indicated that the product was the proton disolvate, H(CO2)2+. Thus, this carborane acid protonates CO2, while traditional superacids do not, and can be considered as the strongest acid known.
- The Strongest Acid: Protonation of Carbon Dioxide,
Steven Cummings, Hrant P. Hratchian, Christopher A. Reed,
Angew. Chem. Int. Ed. 2016.
DOI: 10.1002/anie.201509425
The carborane H(CHB11F11) was reported to be the strongest acid known, as revealed by its protonation of carbon dioxide to form the proton disolvate H(CO2)2+.

Find more world records from all branches of chemistry on the Records and Challenges platform of The Chemical Record.