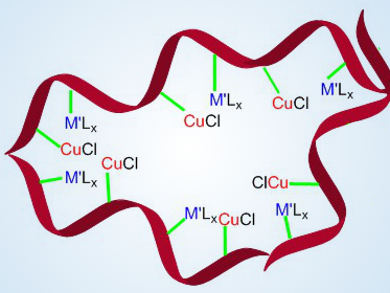
Linking different metal complexes to polymers is an attractive method for the preparation of novel bifunctional catalysts, which can be easily recovered. Unfortunately, such compounds are difficult to prepare by a post-synthesis strategy involving metalation after polymer synthesis.
Fuwei Li, Chinese Academy of Sciences, Lanzhou, China, F. Ekkehardt Hahn, University of Münster, Germany, and colleagues have prepared strictly alternating metallized polymers (pictured). They achieved this by using a click reaction between mononuclear metal complexes (secondary building units, SBUs) bearing N-heterocyclic carbenes (NHCs) functionalized with either p-azidophenyl or p-ethynylphenyl groups. The linker between the SBUs can be flexible or rigid, which influences the catalytic activity of the heterobimetallic compounds.
The researchers observed a high catalytic activity in the alkynylation of trifluoromethyl ketones to give fluorinated propargylic alcohols using a flexible methylene triazole bridge between the metal complexes. The team is currently studying the synthesis of new heterobimetallic tagged molecular compounds and polymers by click-linkage of suitable SBUs.
- Modular “Click” Preparation of Bifunctional Polymeric Heterometallic Catalysts,
Wenlong Wang, Liyuan Zhao, Hui Lv, Guodong Zhang, Chungu Xia, F. Ekkehardt Hahn, Fuwei Li,
Angew. Chem. Int. Ed. 2016.
DOI: 10.1002/anie.201600999