Solar Cells That Generate Power When It Rains
Solar energy is on the rise. Many technical advances have made solar cells quite efficient and affordable in recent years. A big disadvantage remains in the fact that solar cells produce no power when it’s raining. This may change, however: In the journal Angewandte Chemie, Chinese researchers have now introduced a new approach for making an all-weather solar cell that is triggered by both sunlight and raindrops.
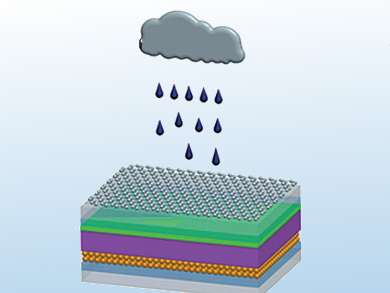
For the conversion of solar energy to electricity, the team from the Ocean University of China, Qingdao, and Yunnan Normal University, Kunming, China, developed a highly efficient dye-sensitized solar cell. In order to allow rain to produce electricity as well, they coated this cell with a whisper-thin film of graphene.
Graphene Electrodes Use the Power of Raindrops
Graphene is a two-dimensional form of carbon in which the atoms are bonded into a honeycomb arrangement. It can readily be prepared by the oxidation, exfoliation, and subsequent reduction of graphite. Graphene is characterized by its unusual electronic properties: It conducts electricity and is rich in electrons that can move freely across the entire layer (delocalized). In aqueous solution, graphene can bind positively charged ions with its electrons (Lewis acid-base interaction). This property is used in graphene-based processes to remove lead ions and organic dyes from solutions.
This phenomenon inspired researchers working with Qunwei Tang to use graphene electrodes to obtain power from the impact of raindrops. Raindrops are not pure water. They contain salts that dissociate into positive and negative ions. The positively charged ions, including sodium, calcium, and ammonium ions, can bind to the graphene surface. At the point of contact between the raindrop and the graphene, the water becomes enriched in positive ions and the graphene becomes enriched in delocalized electrons. This results in a double-layer made of electrons and positively charged ions, a feature known as a pseudocapacitor. The difference in potential associated with this phenomenon is sufficient to produce a voltage and current.
- A Solar Cell That Is Triggered by Sun and Rain,
Qunwei Tang, Xiaopeng Wang, Peizhi Yang, Benlin He,
Angew. Chem. Int. Ed. 2016.
DOI: 10.1002/anie.201602114