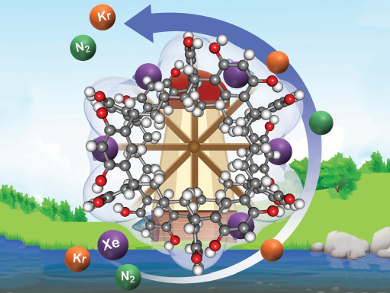
Isotopes of the noble gases xenon and krypton are commonly found in ppm quantities in the off-gas of used nuclear fuel (UNF) from power plants. Capture and separation of these elements is of concern to industry and the environment. The established method by which to do this is cryogenic distillation, which, unfortunately, is very energy intensive and leads to explosion hazards. Moreover, xenon can be used for lighting, imaging, and anesthesia, so its purification costs could be offset in the chemical market.
Praveen K. Thallapally, Pacific Northwest National Laboratory, Richland, WA, USA, and colleagues have developed a much less energy-intensive physisorption-based technique to separate Xe from Xe/Kr mixtures in UNF off-gas. Using acid-catalyzed condensation of resorcinol or pyrogallol and glutyraldehyde, they synthesized the crystalline porous organic oligomeric cage-like materials noria (from resorcinol) and PgC-noria (from pyrogallol) as adsorbents for Xe and Kr capture. As Xe has a larger electron cloud, it outcompetes Kr for adsorption sites in the porous materials.
Noria shows excellent Xe selectivity under dilute conditions, as well as high thermal stability, rendering it a promising candidate for industrial Xe capture.
- Noria: A Highly Xe-Selective Nanoporous Organic Solid,
Rahul S. Patil, Debasis Banerjee, Cory M. Simon, Jerry L. Atwood, Praveen K. Thallapally,
Chem. Eur. J. 2016.
DOI: 10.1002/chem.201602131