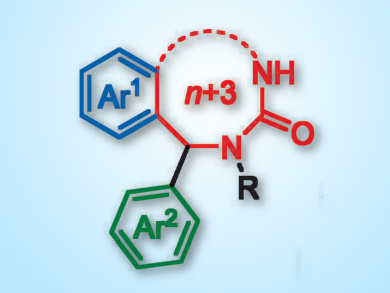
More than 80 % of approved drug molecules contain a nitrogen heterocycle within their structure. Almost all of these are easy-to-make 5–7 membered rings. Although medium-ring nitrogen heterocycles (containing 8–12 members) are present in biologically active natural products, they are absent from medicinally important structures. This is principally due to the difficulties associated with their synthesis. Common cyclization methods are notoriously inefficient and the most common methods employ expensive metal catalysts, thus limiting their industrial use.
Jonathan Clayden, University of Bristol, UK, and colleagues have developed a simple and efficient method for the construction of medium-ring benzo-fused nitrogen heterocycles by using a migratory ring expansion. Benzylic urea derivatives of benzo-fused nitrogen heterocycles undergo this expansion after deprotonation with lithium diisopropylamide (LDA) in the presence of N,N’-dimethylpropylideneurea (DMPU). The reaction allows the synthesis of an array of medium-ring structures in moderate to excellent yields (49–90 %), with excellent enantio- and diastereoselectivity.
This method can be scaled up to synthetically useful levels and allows rapid access to some complex molecular architectures. The ability to make medium rings simply and efficiently could facilitate the investigation of medicinal chemistry of this unexplored region of chemical space.
- Medium-Ring Nitrogen Heterocycles through Migratory Ring Expansion of Metalated Ureas,
Jessica E. Hall, Johnathan V. Matlock, John W. Ward, Katharine V. Gray, Jonathan Clayden,
Angew. Chem. Int. Ed. 2016.
DOI: 10.1002/anie.201605714