With great (nuclear) power comes great responsibility and in the case of exposure to humans or the environment, effective ion chelation methods are required to sequester the harmful agents.
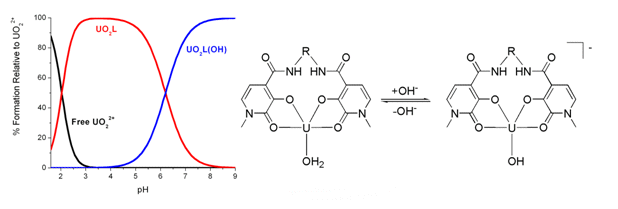
Raymond and Szigethy, University of Berkley, CA, USA, report the design and synthesis of biologically-inspired, water-soluble, uranyl cation extraction ligands. A triethyleneglycol moiety (R, see scheme) increased the water solubility of the ligands, which were then assessed for uranyl cation affinity by using solution-phase titration at a range of pHs. Most industrial extraction agents only work at lower pHs, yet these water-soluble agents work at higher pHs as well, allowing them to be used for in vivo applications.
The butyl-linked ligand was found to have the highest affinity at all pH, but no trends were identified in relation to ligand linker length or geometry. Further evaluation to determine their selectivity is ongoing.
- The Influence of Linker Geometry in Bis(3-hydroxyl-N-methylpyridin-2-one) Ligands on Solution-Phase Uranyl Affinity
G. Szigethy, K. Raymond,
Chem. Eur. J. 2011.
DOI: 10.1002/chem.201002372




