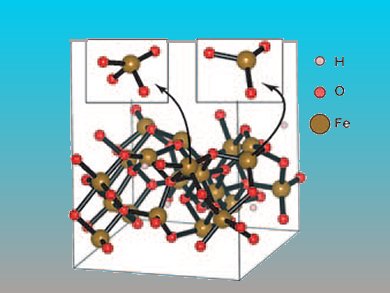
The removal of contaminants from air can be achieved with by mesoporous transition-metal oxides, such as Cr2O3 and Au/MnO2. These oxides are expensive and not environmentally benign. Iron-based oxides such as ferrihydrite (Fh) could replace them if a stable, dehydroxylated form with accessible mesopores can be formed.
Thomas Mathew and colleagues, Toyota Central R&D Labs, Japan, have found that Fh nanoparticles can be precipitated from iron nitrate and NH3. Combined use of 1-propanol and a surfactant gave a cooperative effect that efficiently organized the Fh nanoparticles into a mesoporous assembly which could be partially dehydroxylated by heating.
The mesoporous Fh structure was shown to remove 84 % of CH3CHO at room temperature in 1 h and complete removal was achieved in 3 h. This environmentally friendly material is, therefore, the most efficient iron candidate for complete elimination of organic contaminants in air.
- Mesoporous 2-Line Ferrihydrite by a Solution-Phase Cooperative Assembly Process for Removal of Organic Contaminants in Air
T. Mathew, K. Suzuki, Y. Nagai, T. Nonaka, Y. Ikuta, N. Takahashi, N. Suzuki, H. Shinjoh,
Chem. Eur. J. 2011.
DOI: 10.1002/chem.201002920