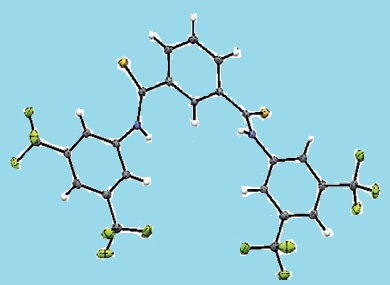
The recognition of anions by host molecules with functional groups such as indoles, pyrroles, sulfonamides, ureas, and amides is important in organocatalysis: hydrogen bonding and electrophile activation play key mechanistic roles.
Rather than synthesizing transition state analogues for target anion receptors, Claire Lennon and co-workers, Waterford Institute of Technology, Ireland, have studied the equilibrium binding of anions to diamides as a mechanistic probe. The diamides were evaluated as anion receptors and organocatalysts in the Baylis–Hillman reaction by NMR titration experiments.
An enhancement of hydrogen bonding was seen with significant increases in catalytic activity. A large degree of pre-organization was observed in one receptor through incorporation of bis-(trifluoromethyl)aniline groups along with a thioamide functionality. The anion binding constants of the series provided a useful tool to probe the catalytic activity of the receptors.
Image: (c) Wiley-VCH
- Synthesis and NMR Binding Studies towards Rational Design of a Series of Electron-Withdrawing Diamide Receptors/Organocatalysts
M. Kinsella, P. G. Duggan, J. Muldoon, K. S. Eccles, S. E. Lawrence, C. M. Lennon,
Eur. J. Org. Chem. 2011.
DOI: 10.1002/ejoc.201001439