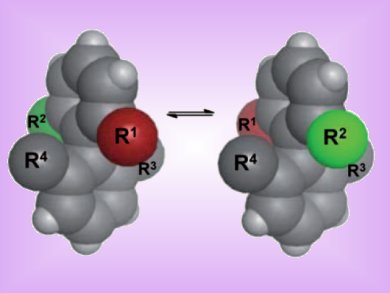
Chirality is particularly important in drug discovery, as separate enantiomers of a given compound can elicit vastly different effects. Steven LaPlante, Oliver Hucke, and fellow researchers at Boehringer Ingelheim in Laval, Quebec, Canada, have highlighted the importance of an often overlooked source of chirality: atropisomerism.
Atropisomers are enantiomers or diastereoisomers that, by slow axial rotation, can interconvert thermally, but for which the half-life of interconversion is 1000 s or more, thus allowing analytical separation.
LaPlante and colleagues used a set of test compounds representing a wide range of rotational energy barriers and corresponding interconversion half-lives (0.002 s to more than ten years) in order to generate a method by which atropisomerism can be predicted. Rotation about the sterically hindered axial bond of interest was simulated by quantum mechanical calculations. They then obtained a torsion profile by plotting the 24 resulting energy values against the dihedral angle. From these torsion profiles, the lowest energy barrier permitting interconversion between the geometric isomers was determined for each compound in the validation set. The researchers observed good correlations between the calculated energy barriers and published data, suggesting that their method is well suited for estimating the barriers to rotation of sterically hindered single bonds and thus for evaluating the likelihood of forming atropisomers.
- Revealing Atropisomer Axial Chirality in Drug Discovery
S. R. LaPlante, P. J. Edwards, L. D. Fader, A. Jakalian, O. Hucke,
ChemMedChem 2011, 6(03).
DOI: 10.1002/cmdc.201000485