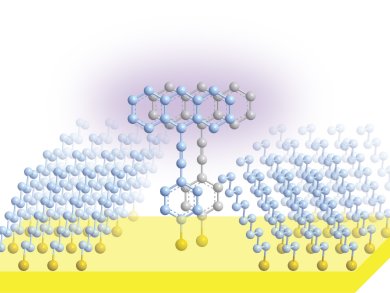
Paul Weiss and colleagues, University of California, Los Angeles, CA, USA, have demonstrated a method for isolating two molecules on a substrate and controlling how they react when excited with ultraviolet light. This technique could allow access to conjugated organic molecules allowed by selection rules but unobserved in solution due to unfavorable reaction geometries.
The technique involved a defect in a self-assembled monolayer (SAM) on a gold surface. This defect was designed so that two sulfur-modified organic molecules would attach in the correct alignment prior to reaction. UV light was used to initiate the photochemical transformation which could be monitored by scanning tunneling microscopy. A [4+2] Diels-Alder addition reaction between the ethynyl moiety and the central ring of a second 9-phenylethynylanthracene could be triggered in this way, rather than the more common [4+4] addition.
- Creating Favorable Geometries for Directing Organic Photoreactions in Alkanethiolate Monolayers
M. Kim, J. N. Hohman, Y. Cao, K. N. Houk, H. Ma, A. K.-Y. Jen, P. S. Weiss,
Science 2011, 331 (6022), 1312–1315.
DOI: 10.1126/science.1200830