Catalytic carbonylation is a common process for the production of carbonyl compounds. There are several examples for the insertion of carbon monoxide (CO) into C(sp2)–H bonds. In contrast, the carbonylation of C(sp3)–H bonds is rare and and has limited substrate scopes.
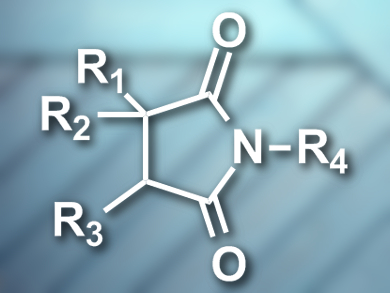
Basker Sundararaju and colleagues, Indian Institute of Technology, Kanpur, have developed the first regioselective cobalt-catalyzed carbonylation of unactivated internal and terminal C(sp3)–H bonds. The team used Co(acac)2 (acac = acetylacetonate) as a catalyst, sodium carboxylate and silver carbonate as oxidants, and 8-aminoquinoline as an auxiliary group. They were able to synthesize a range of succinimide derivatives (example pictured) in good yields.
The proposed reaction mechanism involves the oxidation of Co(II) to Co(III), coordination of the auxiliary group to Co, deprotonation and formation of cobaltacycle, insertion of CO, and a reductive elimination to release the product. According to the researchers, The spproach could be useful for the synthesis of new biologically active compounds.
- Cobalt catalyzed carbonylation of unactivated C(sp3)–H bonds,
Nagaraju Barsu, Shyam Kumar Bolli, Basker Sundararaju,
Chem. Sci. 2017.
DOI: 10.1039/c6sc05026c