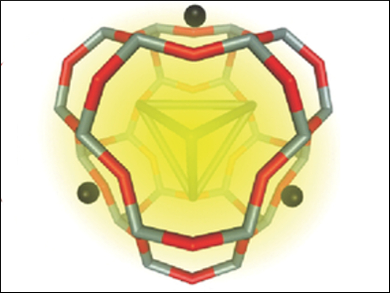
Luminescent silver metal clusters with well-defined properties can be synthesized inside the channels of zeolites. Such composite materials are interesting because zeolites can already be produced on industrial scales. Furthermore, stabilization of the Ag clusters in the zeolite matrix may offer more stable emissive properties than in conventional phosphors.
Mark Van der Auweraer, Marten Roeffaers, Katholieke Universiteit Leuven, Belgium, and colleagues have prepared the first composite inorganic/organic LED based on silver zeolites, dispersed in polyvinyl carbazole (PVK) as the charge transporting layer. Ag zeolites with an Ag/Na ratio of 6/12 (pictured) were prepared via ion exchange of a commercially available zeolite. The team immersed the zeolite powder in an aqueous silver nitrate solution, filtered the suspension, and calcined the powder at up to 450 °C. The resulting composite material showed photoluminescence bands around 550 and 600 nm, depending on the excitation wavelength. When the materials were embedded in PVK, this photoluminescence disappeared but electroluminescence was observed.
The team could tune these electroluminescent properties by varying the Na/Ag ratio in their materials, thereby achieving emission from red to blue light and even emission of near-white light. The researchers hypothesize that PVK only plays a minor role in charge injection and transport. This might lead to emitters that circumvent the inherent instability of organic molecules.
- Silver Zeolite Composites-Based LEDs: A Novel Solid-State Lighting Approach,
Koen Kennes, Eduardo Coutino-Gonzalez, Cristina Martin, Wouter Baekelant, Maarten B. J. Roeffaers, Mark Van der Auweraer,
Adv. Funct. Mater. 2017.
DOI: 10.1002/adfm.201606411