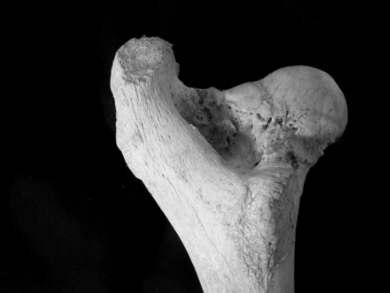
Orthopedic implants have the drawback of being permanent, meaning they either have to be removed via surgery or are left in the body indefinitely. To date, no bioresorbable implants exist that fulfill the requirements of maintaining high strength for support, whilst being biocompatible and degradable within a suitable period of time, usually between six months and two years.
Michael Heiden, Purdue University, West Lafayette, USA, and colleagues have analyzed the effect of porosity of Fe–Mn alloys on the degradation process, while immersed in hard tissue environment along with bone marrow stromal cells. The team used the space holder method of NaCl leaching to create a wide range of pore sizes and shapes. For this method salt crystals are put into a mold. Polymer poured over the salt penetrates into the small spaces between the salt crystals. Salt-leaching has been successfully used in the past to construct porous materials, but has not been used toward degradable Fe–Mn alloys.
The researchers found that alloys with pore sizes of 300 µm are more conducive to cell attachment, proliferation, and bone-like apatite formation and have generally better mechanical properties than their counterparts with pores with diameters of 50 µm. The addition of hydroxyapatite into the matrix of the Fe-Mn alloys shows further benefits for the corrosion susceptibility. However, further research is needed to establish the exact right balance between degradability and mechanical strength.
- Bioresorbable Fe-Mn and Fe-Mn-HA Materials for Orthopedic Implantation: Enhancing Degradation through Porosity Control,
Michael Heiden, Eric Nauman, Lia Stanciu,
Adv. Healthcare Mater. 2017.
DOI: 10.1002/adhm.201700120