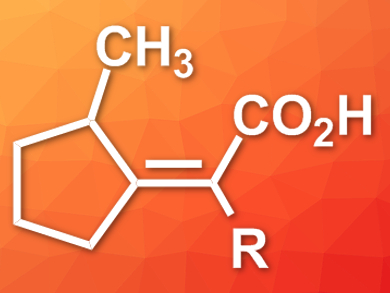
Carbon dioxide is a sustainable chemical feedstock and can be incorporated into organic molecules to form carboxylic acids and their derivatives. Unsaturated carboxylic acids are particularly useful for further functionalization.
Tianning Diao, New York University, USA, and colleagues have developed a nickel-catalyzed coupling of CO2 to enynes that gives α,β-unsaturated carboxylic acids (example product pictured). The team used 1,6- or 1,7-enynes as substrates, a nickel 1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene complex as the catalyst, and diethylzinc as a reductant under 1 atm CO2 pressure to synthesize the unsaturated carboxylic acids.
The reaction selectively gives Z-olefins and tolerates heteroatoms, which allows the synthesis of, e.g., pyrrolidine and furan derivatives. When terminal 1,7-enynes or alkenes with large substituents are used as substrates, the regioselectivity of the reaction changes and the products are unconjugated carboxylic acids. The researchers propose a hydride insertion mechanism for the reaction, which stands in contrast to a transmetalation mechanisms involved in previous reactions that incorporate CO2 into organic compounds.
- Nickel-Catalyzed Reductive Cycloisomerization of Enynes with CO2,
Justin B. Diccianni, Tyler Heitmann, Tianning Diao,
J. Org. Chem. 2017.
DOI: 10.1021/acs.joc.7b01034