Direct formic acid fuel cells (DFAFCs) are a promising new and safe power source as formic acid has a high energy density and is non-flammable at room temperature. In DFAFCs the formic acid is converted into CO2 and water to produce energy: Formic acid oxidation reaction (FAOR) takes place at the anode on a catalytic layer. CO2 is formed and protons pass through a membrane to react with oxygen on another catalyst layer located on the cathode to produce water. The electrons pass through an external circuit from the anode to the cathode and provide electrical power.
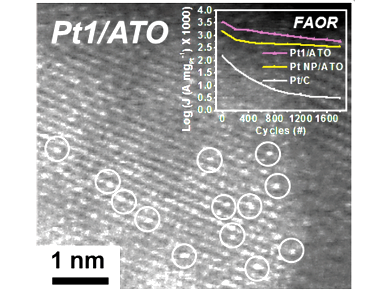
Jiwhan Kim, Korea Advanced Institute of Science and Technology, Daejeon, South Korea, and colleagues have designed a single atomic platinum catalyst supported on antimony-doped tin oxide (Pt1/ATO) for the FAOR. The single atomic Pt structure follows the direct pathway of dehydrogenation of formic acid rather than the indirect pathway via a CO intermediate.
As single metal atoms are usually unstable and prone to aggregation, the team tested the Pt1/ATO catalyst in a DFAFC and found it to be more stable than commercial catalysts for DFAFCs. Additionally, the measured electrical power was an order of magnitude higher for the Pt1/ATO compared to the conventional Pt/C catalysts.
- Highly Durable Platinum Single-Atom Alloy Catalyst for Electrochemical Reactions,
Jiwhan Kim, Chi-Woo Roh, Suman Kalyan Sahoo, Sungeun Yang, Junemin Bae, Jeong Woo Han, Hyunjoo Lee,
Adv. Energy Mater. 2017.
DOI: 10.1002/aenm.201701476