Amyotrophic lateral sclerosis (ALS) (also called motor neurone disease (MND), or Lou Gehrig’s disease) causes the death of neurons controlling voluntary muscles. One of the common causes of ALS is mutations in the superoxide dismutase-1 gene (SOD1) that cause it to misfold and aggregate. The misfolded SOD1 has a reduced capacity for binding metal cofactors, forming a functional homodimer, or partnering with the copper chaperone that activates it. To date, no ALS drugs have specifically targeted SOD1, so identifying such a molecule could represent a breakthrough treatment.
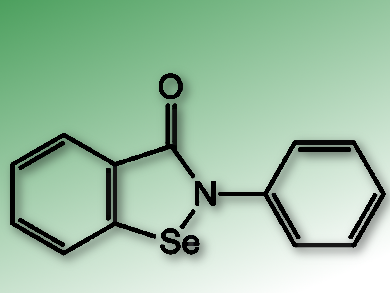
Ebselen (pictured) is a simple organoselenium molecule which has previously been identified and tested for its anti-oxidant and -inflammatory properties. A small screen of cysteine-reactive compounds lead S. Samar Hasnain and his group, University of Liverpool, UK, to identify ebselen for its covalent interaction with SOD1. Subsequently, a crystal structure of the enzyme confirmed that ebselen reacts with Cys111 and helps stabilize the homodimer through ππ stacking with another molecule of ebselen.
What was even more promising is that ebselen was found to greatly increase the affinity of mutant monomers for dimer formation. These SOD1 mutations are known to be important for ALS development and progression and this demonstrates that ebselen could be an important new drug for these patients. In addition, ebselen binding was found not to interfere with the important interaction of SOD1 with its copper chaperone protein. Initial experiments in living cells showed that ebselen could stabilize mutant SOD1, increasing the amount of functional protein by up to six times.
According to the researchers, their results need to be expanded upon, but they hope that a new drug for ALS is just around the corner.
- The cysteine-reactive small molecule ebselen facilitates effective SOD1 maturation,
Michael J. Capper, Gareth S. A. Wright, Letizia Barbieri, Enrico Luchinat, Eleonora Mercatelli, Luke McAlary, Justin J. Yerbury, Paul M. O’Neill, Svetlana V. Antonyuk, Lucia Banci, S. Samar Hasnain,
Nature Communic. 2018.
https://doi.org/10.1038/s41467-018-04114-x