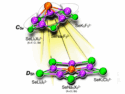
Lithium forms a range of different compounds with both gold and silver at normal atmospheric pressure. Lithium and copper, in contrast, are unreactive and there had been no known binary compounds of the two elements.
Ross T. Howie, Center for High Pressure Science & Technology Advanced Research (HPSTAR), Shanghai, China, and colleagues have studied the reactivity of Li and Cu under pressure. The team used diamond anvil cells (DACs) to compress mixtures of the two elements and X-ray diffraction to characterize the products. The researchers found the first compounds of lithium and copper: At pressures of around 1 GPa, a layered LiCu phase was formed, and at higher pressures of > 5 GPa, a Li2Cu phase with linear chains of Cu atoms could be detected.
These findings are a demonstration of the large effect even relatively modest pressures can have on reactivity. They are also interesting in light of the fact that copper has been used as an electrode material to measure the conductivity of lithium under pressure due to the belief that the two metals do not react.
- Enhanced Reactivity of Lithium and Copper at High Pressure,
Jack Binns, Philip Dalladay-Simpson, Mengnan Wang, Eugene Gregoryanz, Ross T. Howie,
J. Phys. Chem. Lett. 2018.
https://doi.org/10.1021/acs.jpclett.8b01350