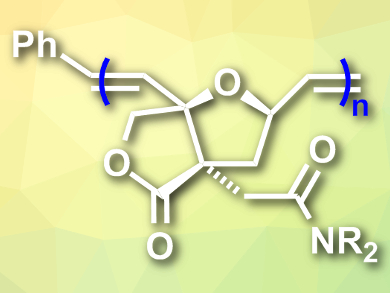
Michael North and colleagues, University of York, UK, have used a ring-opening metathesis polymerization (ROMP) to polymerize biobased oxanorbornene amides and prepare sustainable polymers (pictured). The team used a Diels-Alder cycloaddition to combine the sustainably sourced chemicals itaconic acid and furfuryl alcohol. This reaction was followed by a lactonization to give an oxanorbornene lactone. This product has a carboxylic acid substituent that was converted to a range of tertiary amides using oxalyl chloride, dimethylformamide (DMF), and disubstituted amines in CH2Cl2. The resulting nitrogen-functionalized monomers were dissolved in CH2Cl2 and combined with a ruthenium-based second-generation Grubbs metathesis catalyst to initiate the polymerization.
Using different amines to modify the monomers, the researchers prepared eleven different amides. From these monomers, they obtained a variety of homo- and copolymers with controlled molecular weights. The polymers are amorphous and most of them have high decomposition temperatures.
- Ring-Opening Metathesis Polymerization of Tertiary Amide Monomers Derived from a Biobased Oxanorbornene,
Stefan B. Lawrenson, Sam Hart, Ian D. V. Ingram, Michael North, Rachel R. Parker, Adrian C. Whitwood,
ACS Sustainable Chem. Eng. 2018.
https://doi.org/10.1021/acssuschemeng.8b00779


![Synthesis of [c2]Daisy Chains via Mechanochemistry](https://www.chemistryviews.org/wp-content/uploads/2025/04/202504_RotaxanesWithSolidStateMechanochemistry-125x94.png)