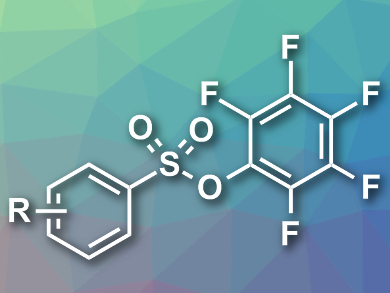
Functional groups that are derived from sulfonyl compounds are useful in organic chemistry. However, sulfonyl chlorides, which are usually used as reagents to form such groups, are very reactive and, thus, sometimes difficult to handle and store. Pentafluorophenyl sulfonate esters (pictured) could potentially be used as sulfonyl chloride mimics, but their synthesis usually requires sulfonic acids with limited commercial availability.
Michael C. Willis, University of Oxford, UK, and colleagues have developed an alternative synthesis of pentafluorophenyl sulfonate esters. The team used aryl boronic acids as starting materials, DABSO (1,4-diazabicyclo[2.2.2]octane bis(sulfur dioxide)) as an SO2 source, and Pd(OAc)2 as a catalyst to prepare sulfinate intermediates. These were then converted to the desired sulfonate esters using pentafluorophenol, Na2S2O8 as an oxidant, and CuBr2 as a catalyst.
The reaction tolerates a wide range of aryl and heteroaryl boronic acids and gives the desired sulfonate esters in good yields. The team showed that aryl bromides are also viable substrates for the reaction, however, they required longer reaction times.
- Copper-Catalyzed Synthesis of Activated Sulfonate Esters from Boronic Acids, DABSO, and Pentafluorophenol,
Vincent Vedovato, Eric P. A. Talbot, Michael C. Willis,
Org. Lett. 2018.
https://doi.org/10.1021/acs.orglett.8b02445


![Calix[4]arene “Handshakes” via Urea–Carboxylate interactions](https://www.chemistryviews.org/wp-content/uploads/2024/04/calixarenehandshake_2024-125x94.png)