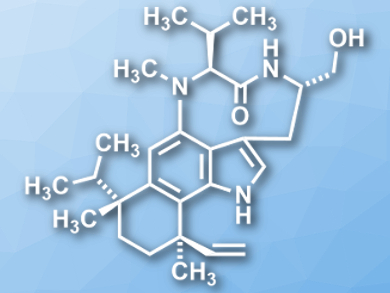
Teleocidins B-1 through B-4 (B−4 pictured) are a family of natural products which were isolated from Streptomyces mecliocidius. The compounds are indole-alkaloids and can activate the enzyme protein kinase-C (PKC). The four different members differ in the configurations at two stereocenters (C19 and C22). Syntheses for teleocidins B-3 and B-4 reported so far require 17−28 steps.
Phil S. Baran and colleagues, The Scripps Research Institute, La Jolla, CA, USA, have developed a simple, eleven-step synthesis of teleocidins B-1 through B-4. The team started from commercially available 4-bromoindole. They used an electrochemical aryl amination at C4 and a Cu-mediated tryptophol construction at C3, followed by a macrolactonization to prepare indolactam V as a key intermediate. This five-step synthesis is the shortest path to indolactam V reported so far.
A C−H borylation allowed the researchers to functionalize the indole at C6. At this point, the synthesis diverges based on the desired product stereoisomer. The team used a stereocontrolled quaternary center formation via a Sigman−Heck reaction to prepare intermediates with the desired configuration at C19. Finally, an addition of vinyllithium followed by a ring closure gave all four desired products.
- 11-Step Total Synthesis of Teleocidins B-1–B-4,
Hugh Nakamura, Kosuke Yasui, Yuzuru Kanda, Phil S. Baran,
J. Am. Chem. Soc. 2019.
https://doi.org/10.1021/jacs.8b13697
Also of Interest
- The Charm and Appeal of Organic Chemistry,
Phil S. Baran, Frauke Zbikowski
Chemviews Mag. 2017.
https://doi.org/10.1002/chemv.201700086
Interview with Phil Baran, who is well known for the synthesis of complex molecules - Dawn of a New Age in Synthetic Organic Electrochemistry,
Chemviews Mag. 2017.
https://doi.org/10.1002/chemv.201700062
Phil S. Baran presented the ElectraSyn 2.0 and demonstrated how easy organic reactions will become - Video: Phil Baran on What Makes a Good Chemist,
ChemistryViews.org 2017.
https://doi.org/10.1002/chemv.201700029
Phil Baran about his research and what makes a good chemist