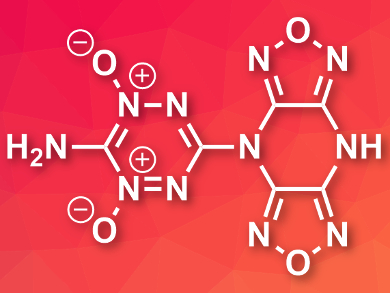
Polycyclic compounds often stack very efficiently in crystals due to interactions between their π-electron systems. This can be useful for the development of energetic materials (explosives, propellants, etc.), which usually require a high density for good performance.
Christopher J. Snyder, David E. Chavez, Los Alamos National Laboratory, NM, USA, and colleagues have developed polycyclic N-oxides based on 1,2,4,5-tetrazine and 4H,8H-difurazano[3,4-b:3′,4′-e]pyrazine (DFP), which can be used as energetic materials. The team performed an addition reaction between DFP as a nucleophile and 3-chloro-6-(3′,5′-dimethylpyrazol-1-yl)-1,2,4,5-tetrazine as an electrophile in different ratios to give 1:2 or 1:1 adducts. These intermediates were then treated with ammonia and finally oxidized using peroxytrifluoroacetic acid (PTFA) to give the desired products (example pictured based on 1:1 adduct).
The synthesized polycyclic N-oxides were found to be less sensitive to impact and friction than the widely used explosive RDX (cyclotrimethylenetrinitramine). They also have excellent thermal stabilities and detonation performances. According to the researchers, the sensitivity and detonation properties of the materials can be tuned by varying the size of the polycyclic N-oxide system.
- Polycyclic N-Oxides: High Performing, Low Sensitivity Energetic Materials,
Christopher Snyder, Lucille Wells, David Chavez, Gregory H. Imler, Damon A. Parrish,
Chem. Commun. 2019.
https://doi.org/10.1039/c8cc09653h